Graphene oxide degradation and dissolution of hydrogel
Combination of chelators: DMSA and EDTA
Legend:
GSH = glutathione
GO = graphene oxide
The following article is based on my previous posts and goes further into the chelation of heavy metals as well as degrading graphene oxide.
I’m documenting my own journey to recovery which is why it should not be considered as medical advice under any circumstances.
1. GENERAL CHEMISTRY OF METALS
In adults about 60% of the body is water, in kids about 75%.
Mn2+, Fe2+, Co2+, Ni2+, Cu2+, Zn2+) have electronegativities in the range 1.6-1.8 => high affinity to electron donor groups containing nitrogen. For example hemoglobin and cobalamine, where Fe2+ and Co2+, respectively are coordinated between four nitrogens. These elements might also bind oxygen, for example, the oxygen-carrying function of hemoglobin. => chelator EDTA
Be2+, Ca2+, and Sr2+ have lower electronegativities => prone to form ionic bonds, for example, to oxygen in the phosphate or carboxylic groups. These elements are often referred to as “hard” or oxygen seeking. => chelator EDTA
Ag+, Au+, Hg2, CH3Hg+, Pd2+, Cd2+, Pt2+, and As3+ are “soft” sulfur-seeking metals and will more easily establish bonds to thiols or selenol groups. => chelator DMSA
2. ESSENTIAL AND NONESSENTIAL ELEMENTS
20 of the elements in the periodic table are defined as essential for humans, with certainty.
4 organic elements H, C, N, and O. 7 “macro-minerals” are essential, namely Na, K, Ca, Mg, Cl, P, and S.
9 trace elements are defined as essential Fe, Mn, Cu, Zn, Se, Co, Ni, Mo, and I.
At present, some other elements are under discussion to be included in the category as essential, such as F, B, Si, and As.
To be categorized as an essential, however, an element must satisfy all of
the following conditions:
It must be present in the human tissues.
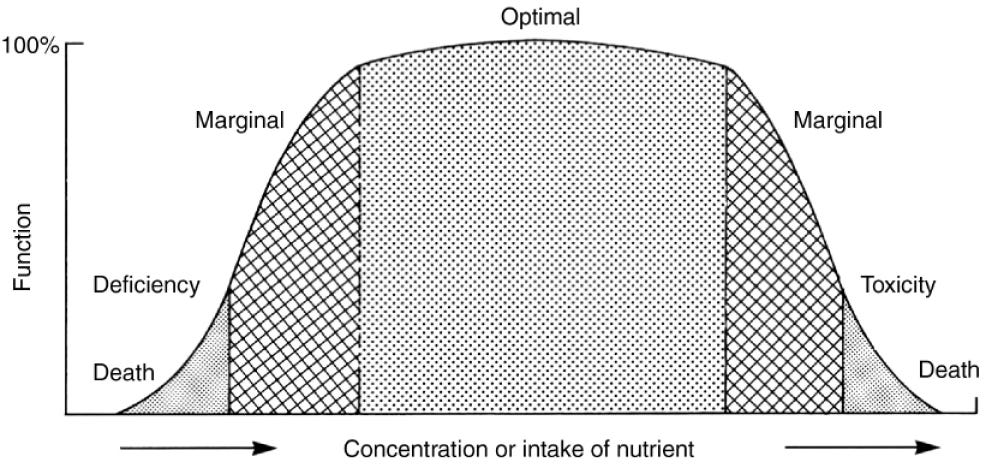
Its dietary deficiency must result in a reduction of a biological function from optimal to suboptimal.
The reduction in physiological function can be normalized by appropriate supplementation of the element (Mertz, 1974).
Note: Some of the readers might be aware of the inclusion of lithium salts by Dr. Nehls to treat Alzheimers and post-vac damage.
Since lithium itself deserves it’s own article, I’m referring to Dr Nehls:
3. EFFECTS OF TOXIC EXPOSURE OF AN ESSENTIAL OR NONESSENTIAL METAL
3.1 Basic Concepts in Chemical Toxicity Testing
Both essential and nonessential metals can become toxic when the amount ingested or the level of exposure goes beyond safe limits. The harmful outcomes that occur at these levels are called critical effects. These effects usually originate in what is known as the critical organ—the specific organ most affected by the toxic substance (Nordberg, 2004). For instance, in the case of exposure to elemental mercury vapor, the central nervous system is considered the critical organ.
Factors for metal toxicity: concentration range, speciation and oxidation state.
Understanding Dose-effect and dose-response relationships is foundational knowledge in toxicology.
Dose-effect relationship => increase in the dose of a chemical (metal compound) causes a quantifiable increase in the toxic effect observed or if additional undesirable effects occur.
Dose-response relationship:
if an observed effect is not quantifiable in single individuals, but is either present or not present (often called all-or-none effect)
if the percentage of a population responding with that effect depends on the dose of the chemical.
3.2 Exposure Patterns and Mechanisms of Metal Toxicity
Factors: Route of absorption; the dose and the chemical and physical form of the metal concerned; genetic variation manifested through racial, familial, and individual susceptibility; nutritional status; immunological status; and presence of intercurrent disease.
3.3 Gastrointestinal Effects of Metal Exposure
Ingesting a toxic amount of metals, usually in the form of soluble salts, can cause acute gastroenteritis. This often happens when acidic foods or drinks dissolve metals from contaminated containers. Symptoms appear soon after ingestion and may affect several people at once, sometimes being mistaken for “viral food poisoning.” Common signs include vomiting and diarrhea, which can progress to circulatory failure and damage to other organs, depending on the metal involved.
3.4 Respiratory Effects of Metal Exposure
Breathing in freshly formed metal fumes can lead to acute chemical pneumonitis, often accompanied by fluid buildup in the lungs (pulmonary edema).
3.5 Hepatic and Renal Effects
Metal poisoning can also cause kidney injury, presenting as a sharp decrease or complete stop in urine production (acute oliguria or anuria) due to tubular necrosis. This kidney damage often develops after earlier gastrointestinal, circulatory, or respiratory symptoms.
3.6 Effects on the Nervous System
Some metals can directly affect the central nervous system, leading to acute neurological illness. Studies show that long-term exposure to household dust containing lead, particularly from old paint, can impair cognitive development in children.
3.7 Hematological Effects
Inhaling arsine (AsH₃) or stibine (SbH₃) gases can cause acute hemolytic anemia—rapid destruction of red blood cells—often combined with kidney failure. This reaction is unique to arsine and stibine, as these gases attack hemoglobin, leading the body to break down red cells.
3.8 Cardiovascular Effects
Certain metal ions disrupt the normal activity of heart muscle cells, leading to abnormal rhythms (arrhythmias), including ventricular fibrillation. Such disturbances can be fatal in poisonings involving antimony, barium, or lithium salts.
3.9 Metal Allergies
Nickel allergy is the most common form of metal-induced skin sensitivity. Alloys containing nickel, cobalt, or palladium are frequent causes of allergic reactions.
3.10 Carcinogenic Effects
Compounds of arsenic, cadmium, chromium, nickel, and beryllium are known to be carcinogenic in humans.
4. PROPERTIES OF CHELATING AGENTS
Crucial qualities of a chelator:
appropriate pharmacokinetics;
high affinity toward the toxic metal;
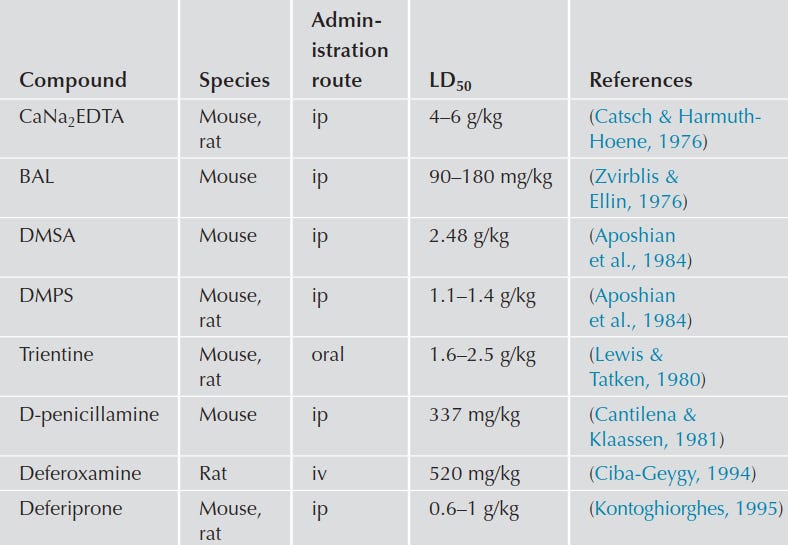
low toxicity;
formation of chelate with rapid elimination (detoxification).
Stable complexes typically form when hard bases interact with hard acids, or soft bases interact with soft acids (HSAB theory by Pearson).
Example: O2 (hard base) and Fe (II) or Fe (III) (hard acid).
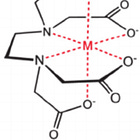
The trivalent iron Fe (III) is classified as a “hard” or oxygen-seeking metal ion. The oxygen in EDTA is a “hard” ligand and therefore EDTA would be the choice for removal of iron (III and II). Moreover, EDTA has 4 carboxyl and 2 aminogroups for binding resulting in very stable metal-EDTA chelate complexes.
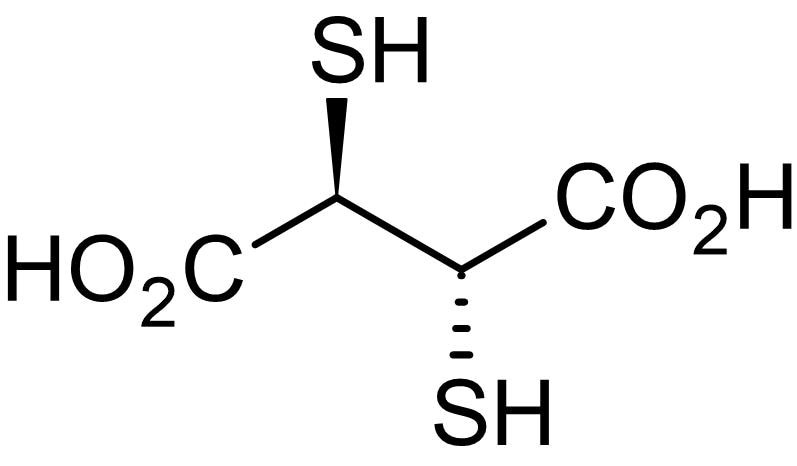
In contrast, ligands that are easily polarized, like those containing sulfur or selenium, are considered soft groups. These form stable complexes with metals like mercury, polonium, arsenic, copper(I), and lead. For instance DMSA (see fig. 2 and 4) does strongly bind to lead and is also FDA approved as an antidote since 1991 to treat heavy metal detoxification (lead, mercury and arsenic for example).
In hard-hard complexes, the bonding is mostly electrostatic (like the bond between iron and oxygen), while in soft-soft complexes, the bonding is more covalent (like the bond between mercury and a thiol group).
4.1 Combination of chelating agents
The combined use of DMSA and CaNa₂EDTA in people with chronic lead poisoning resulted in a greater elimination of lead and better improvement of lead-sensitive biochemical markers compared to treatment with Ca-EDTA alone (Flora, Bhattacharya, & Vijayaraghavan, 1995).
5. My own experience
I had a total of 8 chelating sessions, but I haven’t done any quantitative testing via a mobilization test like this one from my 5th chelation session:
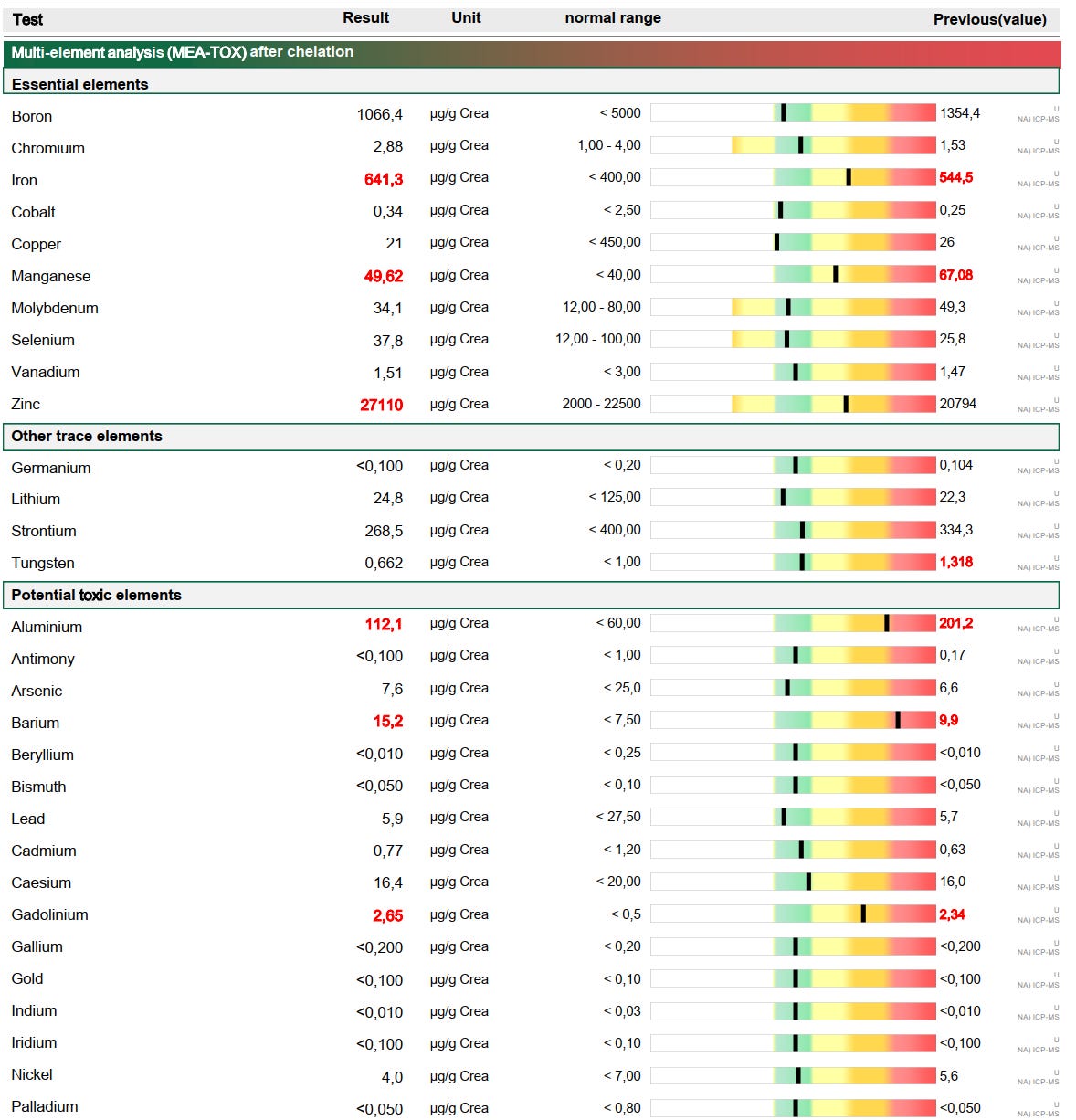
#1-5: CaNa₂EDTA only.
#6: DMSA only.
#7-8: DMSA infusion followed by CaNa₂EDTA.
5.1 DMSA only infusion
The DMSA only infusion was based on my theory that:
DMSA may lead to thiol functionalized graphene oxide (reduced with GSH) via a thiol-ene Michael Addition (click reaction) with a base catalyst (pH > 7). Ignore the ring opening reaction of the epoxide, because it makes this topic too complicated for non-scientists.
(source: https://www.sciencedirect.com/science/article/abs/pii/S245226272100009X)
Assuming that there are also free graphene oxide flakes which are not bound to PEG, a functionalization with thiol groups should render it less toxic, but also make it more biocompatible. The thiol functionalized graphene oxide itself will then become a quite effective heavy metal chelator as several papers are stating this:
Superior Heavy Metal Ion Adsorption Capacity in Aqueous Solution by High-Density Thiol-Functionalized Reduced Graphene Oxides
Adsorption of Functionalized Thiol-Graphene Oxide for Removal of Mercury from Aqueous Solution
Synthesis of amine and thiol dual functionalized graphene oxide for aqueous sequestration of lead
In fact it felt different than just CaNa₂EDTA, based on the urine excretion I’ve witnessed.
5.2 DMSA infusion and subsequent infusion with CaNa₂EDTA
From a heavy metal chelation viewpoint only :
1st step DMSA: Binding heavy metals such mercury, lead, arsenic etc.
2nd step CaNa₂EDTA: Binding heavy metals such as aluminum, iron, manganese etc.
From a post-vax viewpoint only:
1st step DMSA: Render graphene oxide less toxic with DMSA.
2nd step CaNa₂EDTA: Dissolution of the PEG hydrogel with CaNa₂EDTA.
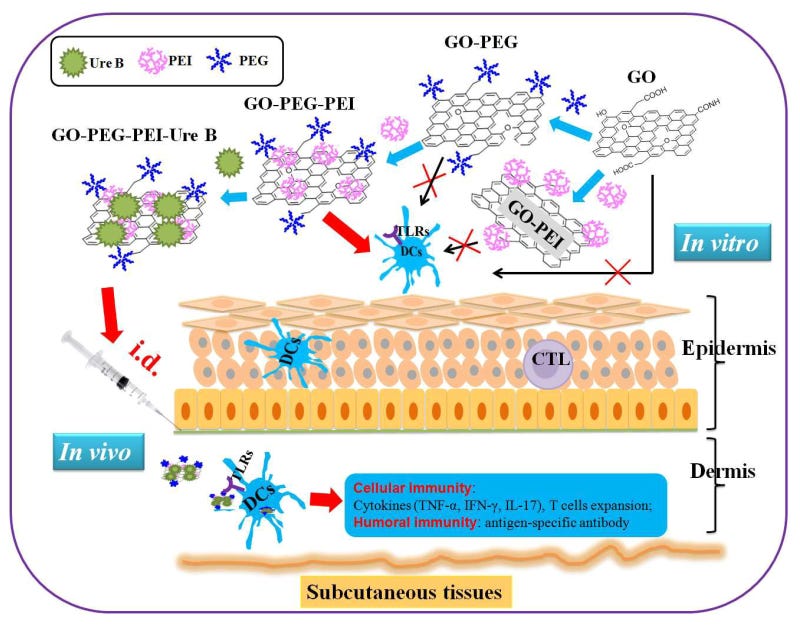
(source: https://pubmed.ncbi.nlm.nih.gov/26814441/)
I also want to remind of you this legendary article by Dr. Ana Mihalcea:
3rd step degradation of graphene oxide:
This topic deserved it’s own post in the future. But there’s only very limited information on how to safely degrade graphene oxide in a human. So here’s what I’ve done so far:
Degradation of graphene oxide with hydrogen peroxide. High dose vitamin c infusions to generate H2O2 (hydrogen peroxide).
I got 2500 mg of vitamin c administered on a separate day, which was not easy as it causes some unpleasant stomach pain.
High-dose vitamin C: A promising anti-tumor agent, insight from mechanisms, clinical research, and challenges
https://linkinghub.elsevier.com/retrieve/pii/S2352304225002314
Degradation of Graphene by Hydrogen Peroxide






I wonder if GO was the cause of "Covid" itself? If that was the case, how was it administered as this was before the "vaccines" were rolled out?
I have been shot up with this biotech/nanotech covertly, via an IV.
And I immediately saw Dr Ana, and had EDTA and a whole bunch of IV infusions that she suggested.
She even admits, that after being injected with this crap, there's no way to remove it, or disable it.
However, she did make my heart palpitations reduce dramatically with her IVs.
Just letting you know....
There appears to be no way out, once you are shot up with this shit.